Summary of Revisions: Standards of Medical Care in Diabetes 2021
American Diabetes Association (ADA) leads in providing clinical recommendations as per the most current state of the field. In addition to many minor changes, the 2021 Standards of Care also contains more significant revisions. The summary of the changes in each section can be studied below:
Objective:
To provide research-based updates that can improve the health and well-being of people with diabetes.
Method:
The guideline was designed with support of currently available evidence and scientific and medical knowledge. The additional changes have been conducted in previous recommendations to clarify recommendations or reflect new evidence.
Recommendations:
Section 1.
In order to improve care and promote health in populations, the information about social determinants of health in diabetes is included, and the “cost-related medication nonadherence” concept has been put into the “Cost Considerations” subsection.
Section 2.
This section focuses on the classification and diagnosis of diabetes. It includes the discussion about the usage of the term LADA (latent autoimmune diabetes in adults) and guidance about the use of A1C assays for diagnosis of diabetes. Additional evidence in subsection “Cystic Fibrosis–Related Diabetes” (CFRD) and “Post transplantation Diabetes Mellitus” is added. A recommendation about screening for diabetes and prediabetes in patients with HIV is shifted to this section from section 4 of previous guidelines.
Section 3.
This section focuses on the prevention or delay of type 2 diabetes. In order to express the increasing awareness and national efforts for healthy lifestyle interventions to prevent diabetes, a new subsection, “Delivery and Dissemination of Lifestyle Behavior Change for Diabetes Prevention,” is introduced in this section. The previous subsection of “Prevention of Cardiovascular Disease” is renamed “Prevention of Vascular Disease and Mortality”. Additional guidance and evidence are been added to this newly named subsection.
Section 4.
This section focuses on comprehensive medical evaluation and assessment of comorbidities. Recommendation 4.6, vaccine-specific recommendations, recommendation on pancreatitis, and HIV are removed from this section. To describe ongoing management, modifications have been conducted in recommendation 4.5 to include overall health status, cardiovascular risk, and risk of hypoglycemia using the risk calculator.
Table 4.1(Centers for Disease Control and Prevention) is introduced in this section that includes detailed information about each vaccine along with important considerations related to COVID-19
Additional evidence for hearing impairment along with audiology for initial care management (“Sensory Impairment” subsection), determining testosterone level (“Low Testosterone in Men” subsection) is been added. Lastly, table 4.1 is been modified to include a number of additional factors, including social determinants of health and identification of surrogate decision-maker and advanced care plan.
Section 5.
This section elaborates on facilitating behavior change and well-being to improve health outcomes. Barriers to diabetes self-management education and support (DSMES) along with ways to address it, 4 critical times DSMES needs should be evaluated and additional evidence on the usefulness of DSMES is added in this section. To address additional guidance and studies, the “Carbohydrates” and “Fats” subsections are being revised. Recommendation 5.29 has been included in the “Physical Activity” subsection in order to encourage diabetic patients for Non-sedentary activities. In order to include smoking cessation as a part of diabetes education programs, recommendation 5.34 is included in this section. Lastly, the concept of mindful self-compassion has been included in the “Diabetes Distress” subsection.
Section 6.
This section elaborates on glycemic targets. Recommendations 6.1 and 6.2 include other glycemic measures other than A1C under a new title “Glycemic Assessment”. Glycemic goals, figure 6.1, and recommendation 6.9 for hypoglycemia assessment is been revised. While recommendation 6.3 is being removed, additional information is added to the “A1C and Microvascular Complications” subsection.
Section 7.
This section elaborates on diabetes technology. Recommendations 7.9–7.13, table 7.3 have been revised, and “professional CGM” is a newly introduced word for “blinded” continuous glucose monitoring (CGM). In new guidelines, recommendations 7.9–7.11 recommend CGM as useful for people with diabetes above other forms of insulin therapy not defined by the type of diabetes or age. Additionally, discussion regarding skin reactions with the use of CGM is also introduced in recommendation 7.14 along with a new discussion on education and training based on new evidence. While the recommendation on examination of insulin injection site was removed, this section now includes revised “Insulin Delivery” subsection. A detailed discussion of recommendation 7.27 regarding inpatient use of devices was shifted to later in the section. The “Inpatient Care” subsection includes a review of the use of CGM in the hospital during the COVID-19 pandemic. Recommendation 7.21 under the “Insulin Pumps” subsection includes discussion on insulin pump use for people with diabetes and older adults. Lastly, the possible benefit of systems that combine technology and online coaching has been included in Recommendation 7.26.
Section 8.
This section describes obesity management for the treatment of type 2 diabetes. Recommendation 8.1 has been updated with the concept of patient-centered communication in addition to the “Assessment” subsection. Health outcomes of weight loss is been discussed in the updated “Diet, Physical Activity, and Behavioral Therapy” subsection. Along with a more detailed “pharmacotherapy” subsection, social determinants of health are being added in this section.
Section 9.
This section discusses pharmacologic approaches to glycemic treatment. The section includes additional evidence-based discussion for the use of sensor-augmented insulin pumps and potential for verbalization with insulin therapy. While table 9.1 is updated, figure 9.1 (decision pathway for CKD and heart failure) and 9.2 (assessment of insulin dose adequacy, use of glucagon-like peptide 1 receptor agonists) is been revised. There is the removal of the concept that improved technologies and treatments with a reconsideration of the role of pancreas and islet transplantation.
Section 10.
This section discusses the cardiovascular disease and risk management. Recent trials regarding cardiovascular risk reduction strategies in patients with type 1 diabetes is been acknowledged in this section. There is the addition of a lower limit for pregnant patients with diabetes and hypertension in recommendation 10.6, the addition of first-line therapy for hypertension in diabetes and coronary artery disease patients in recommendation 10.10, and the addition of ODYSSEY OUTCOMES to the “Combination Therapy for LDL Cholesterol Lowering” subsection. In the “Antiplatelet Agents” subsection, there is an addition of recommendations 10.37 and 10.38 along with new research from THEMIS, COMPASS, THEMIS-PCI, and VOYAGER PAD. New evidence from cardiovascular outcomes trials is been revised in recommendations 10.43–10.47 of the “Cardiovascular Disease” subsection.
“Cardiovascular and Cardiorenal Outcomes Trials of Available Antihyperglycemic Medications Completed After the Issuance of the FDA 2008 Guidelines: DPP-4 Inhibitors,” is the new title for table 10.3A wherein the CAROLINA trial has been added.
“Cardiovascular and Cardiorenal Outcomes Trials of Available Antihyperglycemic Medications Completed After the Issuance of the FDA 2008 Guidelines: GLP-1 Receptor Agonists,” is the new title for table 10.3B wherein the PIONEER-6 trial has been added.
“Cardiovascular and Cardiorenal Outcomes Trials of Available Antihyperglycemic Medications Completed After the Issuance of the FDA 2008 Guidelines: SGLT2 Inhibitors,” is the new title for table 10.3C wherein the CREDENCE and DAPA-HF trials have been added.
Section 11.
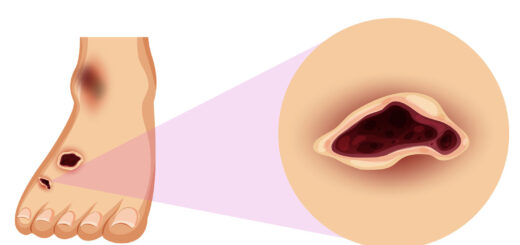
This section discusses microvascular complications and foot care. Recommendation 11.3 is now divided into three recommendations (11.3a, 11.3b, and 11.3c) to segregate treatment based on renal function and risk of cardiovascular disease.
Section 12.
This section discusses older adults. While recommendations 12.4 and 12.5 is been modified in the “Hypoglycemia” subsection, there is the addition of new recommendations on the use of continuous glucose monitoring for decreasing hypoglycemia. Table 12.1 includes modified fasting, bedtime glucose levels, and A1C goal for older adults. Recommendation 12.12 and a review of the evidence on weight loss is added to the “Lifestyle Management” subsection. Table 12.2 in the “Pharmacologic Therapy” subsection is added with reasonable treatment goals for the very complex older patients in poor health.
A1C < 7.0– 7.5% is a modified treatment goal for older adults who are otherwise healthy with few coexisting chronic illnesses and intact cognitive function and functional status.
Lastly, “Incretin-based Therapies” and “Sodium-Glucose Cotransporter 2 Inhibitors” subsections have been added with new considerations and discussion.
Section 13.
This section discusses children and adolescents. A new set of recommendations and their application to treatment decisions has been added to the type 1 (Recommendation 13.12) and type 2 diabetes (Recommendation 13.105) sections in order to incorporate social determinants of health. Three new recommendations have been added to the type 1 diabetes “Glycemic Control” subsection, namely recommendation 13.20 for real-time CGM, recommendation 13.21 for intermittently scanned CGM. At least 60 min daily physical activity with bone and muscle strength training for at least 3 days/week is suggested in updated recommendation 13.58 for youth with prediabetes and type 2 diabetes. Figure 13.1 has been revised to better represent current guidance for management of new-onset diabetes in youth with overweight or obesity with clinical suspicion of type 2 diabetes.
Section 14.
This section discusses the management of diabetes in pregnancy. Understandable information has been updated for insulin requirements during pregnancy in the “Insulin Physiology” subsection. Lower limits have been added for pregnant patients with diabetes and chronic hypertension (recommendation 14.19) and glycemic targets for type 1 and type 2 diabetes in pregnancy. There is an addition of discussion on the use of CGM in pregnancy, especially on time in range and target ranges for pregnant women with type 1 diabetes. Along with new considerations, a guideline on the use of hybrid closed-loop systems during pregnancy has been added. Incomplete data for the use of aspirin in pregnant women with preexisting diabetes is updated along with additional information for its use in the “Preeclampsia and Aspirin” subsection and recommendation 14.18.
Section 15.
This section discusses diabetes care in the hospital. While more guidance on the use of NPH insulin with steroids is updated in the “Glucocorticoid Therapy” subsection, additional information has been added on enteral and parenteral feeding and insulin requirements.
Section 16.
There are no changes made in this section of diabetes advocacy.
Image Credit: Background photo created by xb100 – www.freepik.com